Why Space Is Watery (And So Very Far Away)
In search of heavy water, alien seas and the edge of our solar system
Hello! This is Everything Is Amazing, a word salad newsletter about curiosity, science, attention and wonder.
Alas, Witching Week is over, the New Year has been and gone, and it’s time to reluctantly accept that 2023 is now fully underway. But if you’re finding it hard to let go of the holidays, here’s a little poem by B.J. Novak (Ryan in The Office) that might help you get through the week:
(Hat-tip to Simone Stolzoff of The Article Book Club.)
Secondly, that most welcome of things: an environmental success story! Remember the threat posed by the thinning of the ozone layer in the 1980s?


And according to a report just released by scientists of the United Nations, we’re firmly back on track.
“A report co-produced by UN, US and EU agencies says the Montreal Protocol is working as hoped.
It says that, if current policies are maintained, the ozone layer will be restored to 1980 values - before the ozone hole appeared - at different points in different places:
2066 over the Antarctic, where ozone depletion was the worst
2045 over the Arctic
in about two decades' time everywhere else.”
So it’s official: we can sometimes fix stuff, even the big things.
Ok! Today, in the spirit of tidying up things in preparation for Exciting New Stuff To Come, I’m heading back into the big theme powering the last season of this newsletter - the 71% of our planet we tend to overlook - to ask a question I should have posed at the beginning:
Okay, 71% water. But how much water is that?
This question is prompted by a line from the chapter of Lewis Dartnell’s Origins that some of us have been reading on Threadable:
(That’s not my comment up there - it’s a note left by reader Adam Bodensteiner. Cheers, Adam!)
It’s quite a statement to pause on:
“The water that fills our oceans arrived after the Earth was born.”
I hadn’t stopped to really think about the implications here. But Adam did - and his “wow!” pulled me in, until I was thinking about it properly.
So I started reading into this - and discovered it’s a mindblowing thing to unpack.
On Threadable we’re about to move on to the next text we’re looking at - but it’s not too late to have a read of that chapter of Origins, which you can do for free!
- you’ll need an iPad or iPhone running iOS 12.4 onwards, or macOS 11.0 (apologies, Android & Windows users, Threadable are still in the early development stages here)…
- click the button below:
…after which it should automatically take you to my reading circle, now called “Active Geology with Mike Sowden”.
(If it doesn’t, trying signing in, going to “Home,” finding “All Circles” and swiping sideways until you find it. Request access, and as soon as I see your request, I’ll let you in.)
Now, I might not be the right person for the job here. I’m sure Phil of the Bad Astronomy newsletter or Tad of First Excited State would do a far more competent job, and get all the math correct. Gentlemen, and indeed everyone else, please yell at me if anything that follows is factually awry! Ta.
So here are the questions I tried to answer:
How much water is there on our planet?
Does it really have extraterrestrial origins, as Dartnell claims? (“brought by a bombardment of icy comets and asteroids from the colder, outer regions of the solar system - like a blizzard from deep space”)
How much water is out there, beyond the Earth - and will future astronauts and off-world colonists and explorers ever be in danger of running out of it?
How Much Water Does The Earth Have?
Lacking the time to measure it all by hand, I’m forced to rely on a credible source of data - and few organisations are more credible than the United States Geological Survey, which has been tackling some of the biggest questions in biology, geography, geology, and hydrology over the last 143 years.
It notes that 96.5% of our planet’s water supply is in our oceans. The rest - what we call freshwater (shorthand for “doesn’t contain enough dissolved salts to render it undrinkable”) is held in water vapour, in rivers and lakes and icecaps and glaciers, in the ground (soil moisture & aquifers), and in living creatures, including us humans (we’re 55-60% bags of water, with the remainder comprised mostly of carbon, nitrogen, caffeine, pizza and anxiety).
Since we very quickly sicken and die without adequate hydration, our continued existence as individuals, communities and as a species depends on adequate water supplies. So at first glance these stats are heartening: oh wow, so the tiny fraction of the world’s freshwater that we draw upon is just a vanishingly small part of just 3.5% of the supply we could get from the sea? That’s basically endless!
Well, yes and no. There’s a good reason that we satisfy less than half a percent of human water needs with desalinated water: energy. In a practical sense, it still costs too much to process it. To be able to do so in quantities that truly matter, energy generation (clean energy, to avoid pollutants that end up back in the water supply and render the whole thing pointless) needs to get a lot cheaper.
And - it is! To an quite astonishing degree, as Bill McKibben explained here in July last year.
Nevertheless, in pragmatic terms it’s still mostly impractical at scale. In time, perhaps?
So, uh - that doesn’t look like a lot. Seriously? All the water in that single blue drop?
Firstly, it’s not a drop: it’s a sphere, with a diameter of 860 miles. That means there’s around 332 million cubic miles of water in that thing. Yes, I know that’s just a number, but - you can see how big it is compared to the United States (if it “popped” and emptied all its water onto the U.S., the layer of water would be over a hundred miles deep).
And if it was hovering over Europe, it’d stretch from London to Rome.
A fact I found surprising, via the USGS page:
“Of the freshwater on Earth, much more is stored in the ground than is available in rivers and lakes. More than 2,000,000 mi3 (8,400,000 km3) of freshwater is stored in the Earth, most within one-half mile of the surface.”
So in theory, even if we ran low on freshwater from lakes and rivers - which is frequently the case in many parts of the world, and the subject of a few environmental disasters, like this one - there is still plenty of water locked up not-too-far underground, so if there was an environmentally responsible and cost-efficient way to reach it, we wouldn’t need to rely on processing any seawater to meet our needs?
That’s a lot of “if”s there, and a lot of over-idealistic uses of the word “we”, but - good to know, I think?
Did All Our Water Come From Outer Space?
Dartnell’s statement is solidly on the side of extraterrestrial origins - and it reflects what was until recently the agreed-upon model.
But geologists have been poking at this particular suspiciously-simple-looking model for decades. What’s the actual evidence for such a thing being true?
One indicator is an isotope of hydrogen (deuterium) that goes into making so-called “heavy water” - which is well-named, because if you freeze a sample of it and drop that lump of ice into normal water, it’ll sink to the bottom. The modern ratio of heavy to “light” water on our oceans has the potential to tell a story about where Earth’s water came from (because, for example, many comets seem to be unusually rich with deuterium, so bombardment with similar forms of space-ice could throw that ratio out of whack over time).
It’s an extremely complicated piece of detective-work, but many researchers (like astronomer Karen Meech) are dedicating their careers to untangling it.
Meanwhile, as this 2020 study concluded, water may also have been present in the materials that formed the inner solar system - namely, the stuff that collided and melted together to form the Earth in the first place.
A form of meteorite called an enstatite chondrite has been found to contain enough hydrogen that, if it was of equivalent accumulated size, it could create the water in our planet’s oceans three times over. At first glance this might look like more evidence for extraterrestrial origins, but the hydrogen and nitrogen isotopic compositions of these meteorites also match those of the Earth’s mantle, which has been mostly locked away underground since our planet formed.
It suggests that the vast reservoirs of water under our feet were an innate feature of the material that came together to create the Earth. If true, some of that water - in the form of hydrogen, since the early days of the planet were much too hot to hold water - was here from the start.
Neither model is exclusive of the other. And in the way of these things, a mix of the two seems…not outside the realm of possibility? But no, it doesn’t look as simple as “all our water came from space”. (Sorry, Professor Dartnell.)
Okay, Exactly How Much Water Is Out There?
“Exactly”? Oh come on. Are you trying to give me a headache?
On the one hand, we certainly know that water is all over our solar system. In 2018 the Indian Space Research Organisation’s Chandrayaan-1 lunar probe confirmed the existence of ice in the shadowed areas of the Moon - and in 2020, NASA announced the discovery of water on the sunlit side. (In that latter study’s sample area, it’s roughly enough to fill a 12oz bottle for every cubic metre of lunar soil.)
Further afield, Cassini probe data suggests Saturn’s moon Enceladus has “curtains of vapour” erupting out of cracks in its surface (some of which are up to 75 miles long), suggesting an ocean under the surface that’s up to 6 miles thick.
Plus there’s Europa, with its proposed liquid ocean that descends a truly abyssal 90 miles, as I wrote about previously…
Relatively speaking, our seemingly unremarkable solar system should be pleasantly awash. In 2016, Scientific American writer Shannon Hall reported there’s at least 50 times more water that is found on Earth - and that’s just based on the small amount of confirmed evidence to date.
This isn’t to say it’s going to be easy to tap, as the Moon water example suggests (processing a cubic metre of soil just to get enough water to fill a can of Coke is not what anyone would call “energy-efficient”, let alone “practical”). Nevertheless, technical challenges notwithstanding, our interplanetary future should be more than adequately hydrated.
But I’d like to use all this as a super-thin excuse to talk about my favourite part of our solar system. You know, the really, really insane part.
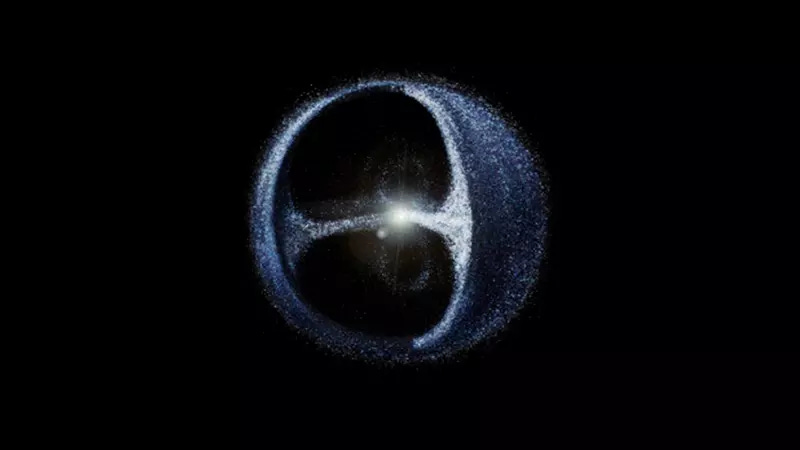
Welcome to the terrifyingly distant edge of our planetary neighbourhood.
There’s no real way to visualise this thing, but here’s one artist’s attempt to at least get the idea of it across in a way that fits into our brains.
The utmost rim of our solar system, more or less everyone agrees, is the point where the Sun’s gravitational pull is negligible compared with that of the other stars in our galactic neighbourhood. And within this boundary should drift an essentially unimaginable amount of fragmented rock and ice, forming billions of comets and trillions of asteroids.
All this stuff drifts around according to the mathematical elegance of the laws of gravitational force. You can see them at work in the image above: an inner “cloud” in the form of a faint torus (or upended infinity-sign, appropriately enough), surrounded by a far larger outer ring in the shape of a scaffolded bubble - or, to my eye, the former Cardassian mining outpost of Deep Space Nine.
It’s called the Oort Cloud, after the Dutch astronomer Jan Oort - and nobody has seen any sign of it yet, even though its existence is not currently in dispute.
As to how far away it is, the best measure is the AU (Astronomical Unit): roughly the distance from the Earth to the Sun, around 93 million miles (150 million km). The inner Oort Cloud begins at 2,000 AU - and its outer edge is anything up to 100,000 AU away.
These are stupid, ridiculous numbers, so here’s another way of thinking of it.
Since 1977, the Voyager 1 space probe has been tirelessly hurtling towards the rim of the solar system. The inner planets and the warm light of our Sun are now far, far behind - and it’s travelling at a speed of over a million miles every day into that profound blackness.
At its current velocity, it’s going to take Voyager another 300 years before it reaches the inner edge of the Oort Cloud - and then it’ll need tens of thousands more years to get out of it again.
Gulp.
But wait - the Oort Cloud is mostly ice? Isn’t that even more water for future explorers to draw upon?
Okay, firstly, you read the bit about how far away it is, right? Just checking. And here’s something that really needs emphasizing: it looks like there is nothing else out there. This is otherwise empty space.
(There was a highly controversial theory that the Oort Cloud contains a gas giant several times the size of Jupiter, called Tyche - a so-called “extra planet” of our solar system - but in 2014, the findings from NASA's Wide-field Infrared Survey Explorer found absolutely nothing to suggest it exists.)
Secondly: the Oort Cloud, in a tangible sense, is itself a whole lot of nothing. We’re talking about a staggeringly vast number of bodies spread out over a space that is itself staggeringly vastly more staggeringly vast. It’s incredibly diffuse. Based on the mass of comets, one estimate for the combined mass of everything within the Oort Cloud is just five times that of Earth. Five Earths, pulverised and scattered over that utterly terrible volume of space…
So no - future astronauts aren’t going to be taking trip to the Oort Cloud to refill their water-bottles. It’s likely no human being will ever see it up close - partly because it’s simultaneously too big and too small to see, and partly because the trip isn’t survivable. Even Voyager 1, when it reaches it in the 24th Century, will be long drained of power. It’s just too far for everything.
Humanity’s best bet is, of course, to look after the plentiful supplies of water it has at home - and learn to share them properly in a spirit of respectful cooperation, assisted by all the new technology we can bring to bear. If we did that, there should be more than enough for everyone.
So hey, maybe we should do that?
Images: USGS; Gatis Marcinkevics; Cristofer Maximilian; Pablo Carlos Budassi/Wikimedia Commons.








You definitely made a splash with this article. I was all in for finding water somewhere far out there until you said it was so far. Guess we'd die of thirst (or something else) before we got there. I'll try taking shorter showers. 💜
“So hey, maybe we should do that?” !!!! Indeed. And if we postulate that the energy that makes up me could be bigger than my body, I tend to think I’m probably 75% anxiety and I don’t know how much of anything else.